Program Overview
In 2016 the U.S. Food and Drug Administration (FDA) established minimum standards for safe growing, harvesting, packing and holding of produce grown for human consumption as part of the Food Safety Modernization Act (FSMA). The Minnesota Department of Agriculture (MDA), under an agreement with the FDA, is building a Produce Safety Program to promote understanding of the new Produce Safety Rule. The Produce Safety Program's mission is to ensure:
All Minnesota produce farmers receive the resources and support they need to grow safe food while meeting any regulatory requirements that apply to their farms.
Steps for Produce Growers
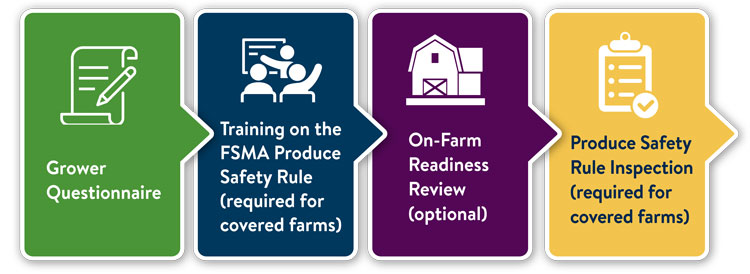
Just beginning to learn about the Produce Safety Rule? Here are some good starting points:
- Learn where your farm falls under the Produce Safety Rule by filling out the FSMA Produce Safety Rule Grower Questionnaire.
- Understand your farm's status and what does or does not apply to your business.
- Take a FSMA Produce Safety Rule Grower Training course. This course meets the training requirement for farms that are covered under the rule.
- Request a non-regulatory On-Farm Readiness Review with MDA and University of Minnesota Extension staff to help identify areas to improve food safety practices on your farm.
- Use the pre-inspection checklist (PDF) to self-identify areas to improve your farm practices.
- Sign up for the Produce Safety Program e-newsletter.
- Learn more about licensing exemptions for produce farms or cottage foods.
Each year we analyze data collected from our annual Grower Questionnaire, evaluations of our grower trainings, and other program services. This data is shared in our annual report which can be found in the forms and resources section on this page.
Farm Inventory
| Farm Produce Safety Rule (PSR) Status Categories | 2024 calendar year |
|---|---|
| Covered | 45 |
| Eligible for an Exemption - Qualified Exemption | 207 |
| Eligible for an Exemption - Commercial Processing Exemption | 61 |
| Excluded - Sales | 797 |
| Excluded - Other | 163 |
| Total Farms in Inventory | 1,273 |
*Verified farms have completed the FMSA Produce Safety Rule Grower Questionnaire, or otherwise verified their farm’s Produce Safety Rule status with the MDA.
Building Safe Farms: Program Impact and Success
We support Minnesota farmers in complying with the FSMA Produce Safety Rule (PSR) through inspections and education. Our team works to ensure safety standards are met while building relationships within the produce community. We prioritize outreach to all types and sizes of farms across Minnesota to help them maintain safe farming practices.
| 44% of our resources were developed in a language other than English |
| 4 Newsletters, totaling to over 5,000 unique opens, were sent out in 2024, each one focusing on a different food safety element. |
51% of all produce farms growing covered produce in the inventory have attended a grower training
|
| 22% of all training held have also been offered in Spanish, Hmong, or accessible for Plain communities (ex. Amish, Mennonite) |
83% of Covered farms under the MDA's inspection responsibility were inspected in 2024
|
Questions?
Have more questions about what the Produce Safety Rule means for you? We want to hear from you! Our team includes a mix of inspectors, outreach staff, analysts, and project specialists. We are all here to support understanding and compliance with the requirements of the rule. Give us a call at 651-539-3648 or send us an email at ProduceSafety.MDA@state.mn.us with any questions you might have.
Funding for this program was made possible, in part, by the Food and Drug Administration through grant PAR-16-137. The views expressed in written materials or publications and by speakers and moderators do not necessarily reflect the official policies of the Department of Health and Human Services; nor does any mention of trade names, commercial practices, or organization imply endorsement by the United States Government.

